Chapter 2
From Muscles to Meals
Allan Lasser
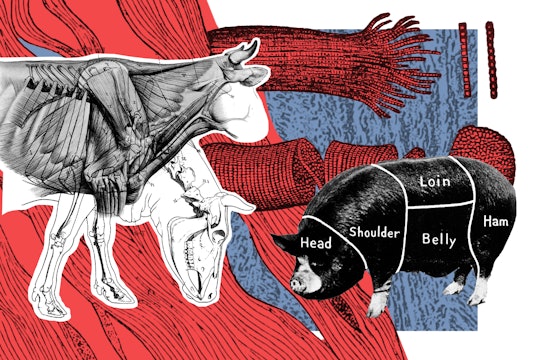
Where does meat come from?
Meat comes from dead animals. But it also comes from industrial processes with complex, invisible supply chains. After its been slaughtered, meat from living animals needs to soften—either by allowing it to sit over time or by treating it with enzymes or physical force. It's transported refrigerated or frozen, inside vacuum sealed bags to prevent bacterial growth and keep the meat attractive for longer.
Despite the growing awareness of the ethical and environmental questions that surround eating animals, the global meat industry is thriving. In the past 55 years, worldwide production of meat increased nearly 5-fold—in 2016, almost 330 million tons of meat were produced globally, compared to 71 million tons in 1961. Almost half of it came from Asia, which became the world's primary meat producer in the early 1990s.
But American businesses make up a hefty percentage of the meat producers. The United States is the world's largest producer of beef and poultry, tallying over 12 million tons of beef and almost 20 million tons of chicken meat products in 2016 alone. The United States is also the largest consumer, the largest importer and the fourth-largest exporter of beef in the world. If you live in the United States, there is a high chance that the chicken, beef, and pork you see in your local grocery store comes from Arkansas and Colorado, home to the two biggest American meat processing companies, JBS USA and Tyson Foods Inc. These giant corporations are involved in both slaughtering and processing, including packaging and distribution. Because these company supply chains have grown so efficient, consumers don't often have to think about what meat is, or how it gets from the farm to their kitchen.

It's easy to overlook that the neat, plastic-wrapped hamburger you find in the freezer aisle begins as an animal. In living animals, a pumping heart supports the circulation of oxygen and nutrients in the blood, allowing the efficient use of carbohydrates to produce the energy used to contract and relax muscles. The circulatory system also ordinarily transports cellular waste away from muscles, preventing its accumulation. But after an animal dies, cells no longer receive oxygen, and a process called anaerobic glycolysis begins. To produce energy, muscle cells break down a stored carbohydrate called glycogen, creating acid as a byproduct. This contracted state of muscle fibers—a permanent stiffness—is called rigor mortis.
Rigor, and its resolution, are very important in meat technology. How and when the rigor develops has a profound effect on the tenderness and juiciness of meat. As the cells acidify, enzymes that break down protein structures activate, weakening the bonds that hold muscle fibers together, softening the meat. That's why it's common for slaughterhouses to store meat for extended periods of time before selling it, a practice known as resolution, conditioning, aging, ripening or maturing. Meat frozen immediately after slaughter doesn't go through this process, and remains considerably tougher.
The amount of time required for optimal tenderizing can vary depending on the type of meat. For example, it takes about four days for pork meat to become noticeably softer and tenderer, but double that for beef, veal, and rabbit. The fast pace of large-scale industrial meat production pushes factories to shorten the time spent conditioning to distribute the product sooner, usually within three days of slaughter. To compensate, factories sometimes choose to further modify the muscle structure with additional processing steps, such as applying physical force, high pressure, electrical stimulation or ultrasonic waves, or by adding salts and plant-derived enzymes to achieve more efficient protein denaturation and tenderer meat.
The availability of refrigeration has also changed the meat industry dramatically. Keeping meat cool helps stop the growth of pathogenic bacteria, preventing it from spoiling and making people sick. Although the muscle of a slaughtered animal is essentially sterile and pathogen-free, the same isn't true of animals' hides and guts. During butchering, there's a high chance of contaminating meat with pathogens naturally present on the surface of animal’s body. This makes prompt refrigeration critical for preventing the growth of dangerous bacteria. Bacteria grow most rapidly in the range of temperatures between 40 and 140°F (4 to 60°C)—so the warmer the meat is, the faster it will spoil.
In the long run, spoilage is essentially inevitable unless meat is frozen solid. Lowering the temperature progressively slows bacterial growth, but doesn't stop it completely. According to early observations, meat develops an off-putting odor in three days when kept at 20 °C, in ten at 5 °C and in 20 days at 0 °C. That's also why it’s important to thaw frozen meat slowly, either in the fridge or in a cold water bath—rapid increase in temperature can reactivate bacteria that were unable to grow in the cold, revamping their growth and becoming a health hazard. Refrigeration and thawing can also impact both water retention and water loss, changing both the quality and look of the product. Generally, the faster the meat is chilled and cut after slaughter, the better it retains water, looking tastier to potential buyers.

After meat is refrigerated and packaged, it's transported to distributors, and from there to grocery stores, butcher shops, and households. While frozen meat has been widely available since the 1930s, better control over storage temperatures now allows chilled “fresh” meat to be shipped long distances. Nowadays, packing meat into a pouch and removing all air using a vacuum chamber is the most commonly used method of transporting meat along the production chain. But while conventional plastic vacuum packaging does extend the shelf life, it comes at the cost of visual appeal. While low oxygen content slows down the growth of certain types of bacteria, it also affects the rate of red meat oxidation—which can make red meat change color from cherry-red to purple-red or grayish-brown. The solution is to use controlled or modified atmospheric packaging (MAP), where, instead of simply removing all gases, gas composition inside a sealed MAP bag is artificially modified to allow meat to stay visually attractive for longer. For packaged red meat, the solution adopted in Norway and Canada is to add carbon monoxide to the mix, which can help improve color stability and prevent red meat from turning brown.
So while you may not realize it, you've probably been breathing in a little puff of laboratory research and innovation every time you open a supermarket pack of chicken thighs or a vacuum-sealed steak.
What is a muscle?
Muscles are an integral component of mammalian skeletons and organ systems that produce movement through directional force. They're highly organized and complex structures, growing from protein fibers into versatile biological machines. There are multiple kinds of muscle cells, but most are comprised of bundles of fat, nerves, and connective tissue. They even have their own personalized stem cells.
As jokes about "meatheads" imply, meat is mostly muscle. But what, exactly, is muscle made of, and how does it perform its function? Though you may not think about it when you tuck into a steak, what you're eating is a specialized cellular hyperstructure which allowed the animal to move while it was alive.
Muscles are extraordinarily complex biological machines. Made of elastic tissue, there are three main types: Cardiac, smooth, and skeletal. Cardiac and smooth muscle groups are not under voluntary control—these muscles are responsible for the contractions in your heart and digestive system, respectively. In contrast, skeletal muscles are the only muscles that can be consciously manipulated. They also happen to be the most important muscle type for meat production.

Skeletal muscles' primary function is to contract, allowing you to make the thousands of small and large movements that get you through your day. To do so, muscles depend on chains of thousands—or tens of thousands—of myofibrils, the basic tube-like unit of a muscle cell. Myofibrils themselves are made from proteins, including actin and myosin, which are organized into thin and thick filaments. When you try to move a muscle, electrochemical signals are sent from the brain to motor neurons. These release a neurotransmitter called acetylcholine, which creates an electric potential across the membrane of the desired muscle's cells, causing calcium ions to flow into them and the filaments to slide. When thick myosin filaments slide along thin actin filament, muscles contract. Coordinating all this to produce movement requires a lot of energy, so muscle cells are also packed with mitochondria, the powerhouse of cells.
Muscles also contain other kinds of tissue. Muscle cells are bundled together and enclosed in a sheath of connective tissue, called the endomysium, which surrounds each individual muscle cell. Woven through the endomysium are nerve fibers and capillaries, which provide sensory feedback and supply the muscles with nutrients from the blood stream. Another common type of structure found in muscles is collagen—an important structural protein which regulates the firmness and elasticity of a given tissue.

As the protein filaments in muscle cells can be very long, muscle cells are organized in an elongated shape. Unlike other cells, muscle cells are produced during a process called myogenesis during neonatal development—which means that, after you're born, you almost never generate new muscle fibers. Instead, existing cells grow in size when the need arises.
Muscle generation begins during embryonic development when stem cells—a unique type of cell that can develop into many types of specialized tissue or organ—start to differentiate into myoblasts, which are the precursor to muscle cells. During myogenesis, some of these myoblasts eventually mature into myobtubes, tubular structures with multiple nuclei (ordinarily, most cells only have one nucleus)—while the rest of the myoblasts continue proliferation to protect against future muscle damage. As muscle-generating myoblasts stop their cell division process and align themselves on a tubular axis, neighboring myoblasts start to fuse their cellular membranes, forming a muscle cell, known as myocytes. Even into adulthood, myosatellite cells (stem cells or myogenic cells, those with capability to make muscle) reside in the skeletal muscles of our bodies. Their most important task is muscle regeneration after damage. For example, if you pull a muscle while exercising, these stem cells can act as building blocks to repair the damage, fusing with the injured muscle cell.

Muscles, of course, can vary widely in size and strength from person to person. That's because muscle cells grow in size in response to mechanical tension, like regular exercise (although the total number of muscle cells stays the same.) This process is called muscle hypertrophy. It's still unclear how this growth is actually achieved, but there are (at least) two hypotheses: The first is known as "sarcoplasmic hypertrophy," which is where an increase in a carbohydrate called glycogen increases the volume of the muscles' storage areas, known as the sarcoplasmic reticulum. This increases the total volume of the muscle. The second is called "myofibrillar hypertrophy," which is where muscle cells try to adapt to external tension—for example, by exercising, which increases the number and size of myofibrils. Most likely, both processes occur in parallel. In livestock, the same rules for muscle growth apply. The more animals move and exercise their muscles, the larger their number of muscular fibrils which may be associated with more juiciness and a stronger meat flavor. (An alternate hypothesis suggests there's no such benefit.) But there's an argument that's why the exercised meat from free-ranging cattle has a stronger flavor and texture than meat from fenced cattle, while cooped up chickens produce a tender meat from muscles which were hardly used during their lives.
How do muscles become meat?
While we think of meat and the animal it comes from as synonymous, muscle tissue undergoes chemical and physical changes before it becomes edible meat. The physical effect of these changes are what scientists are trying to replicate in cultured meats, to create a sensorially palatable imitation of real meat.
Pork, beef, lamb—we tend to closely associate raw meat with the animal it comes from. Yet from the moment of slaughter, irreversible chemical changes take place in the dead animal to create the meals we know.
In fact, the journey from muscle to meat begins even before the slaughtering process. For instance, so-called "pre-slaughter stress" can dramatically affect meat quality. In pigs, this process is referred to as pale soft exudative meat, resulting in meat that is a very pale color and that tastes particularly acidic. In cattle and sheep, the equivalent is the self-explanatory Dark Firm and Dry Meat. Both defects cause the meat to be less valuable and unpopular with consumers. They happens because stress—perhaps caused by handling or stunning—affects the chemistry of the meat after death.

Typically, the reddish color of meat is determined by myoglobin, an oxygen-binding protein in muscle cells, similar to the hemoglobin in your blood. The concentration of myoglobin determines the color of the meat—that's why myoglobin-rich beef is deep red and myglobin-poor poultry characteristically white. When unstressed animals die, lactic acid builds up in the muscle cells, slowing their mitochondria activity. This leaves surplus oxygen (which would normally be consumed by the mitochondria) for myoglobin to bind with. The oxygenated myoglobin then turns a bright red color (think hamburgers). But when stressed animals burn through their glycogen stores, they have none left after death to produce lactic acid. Their mitochondria keep going like gangbusters, using up the muscles' remaining oxygen, and leaving myoglobin unbound—causing the meat to turn an unsightly purplish-pink color. This kind of discoloration costs the U.S. meat industry $1 billion dollars of revenue every year.
Meat undergoes further chemical and structural changes during cooking. As you may have noticed, meat changes both color and texture when warmed, going from red to gray to brown. These color changes are influenced by a few things. What myoglobin—the oxygen-binding protein in your muscles—is doing plays a big role. Myoglobin carries an iron atom which in turn carries oxygen, like your hand carrying a cup which in turn contains tea. Myoglobin that carries a lot of oxygen is a bright pink (like the color of ground beef, which gets mixed with air during grinding). When meat is exposed to air, the iron in myoglobin essentially rusts, spilling its oxygen, causing the meat to turn brown. Heat also denatures meat proteins. When cooked, myoglobin separates from its iron and forms a "hemichrome" pigment, also brown/tan-ish. When myosin and actin, muscle-movement proteins, are cooked, they unfold and form intricate knots—a similar chemical process occurs when you cook an egg. And, like an egg, meat becomes more firm during this denaturation process. After prolonged heat, collagen, the main protein in connective tissue, also breaks down to gelatin, converting cartilage into melting jelly.

Meat's shape is also altered by the loss of water during cooking, which typically causes it to shrink in size. This in itself leads to a drier product—but since fat also melts under heat, the perception of juiciness depends on the cut's proportions of fat and water. In fact, emulsions of fat and water are stabilized by proteins, such as myosin, which are more stable under high temperatures. This higher stability allows more water to remain in fatty meat than in lean meat while cooking. So, a cooked fatty cut of meat will have more water than a lean cut, once both are on the dinner plate.
Beyond these textural changes, meat goes through a series of taste-modifying chemical reactions as temperatures increase. During the Maillard reaction, usually occurring between 140 and 165°C, amino acids (for instance from degraded proteins) react with sugars to form thousands of different compounds, each with its own unique flavor. At higher temperatures, caramelization breaks down the structure of sugars and produces an even greater abundance of flavorful molecules. Finally at extremely high temperatures, pyrolysis, or charring, produces carbon from the complete breakdown of surface molecules. All of these changes contribute to an arguably more complex flavor in cooked food than you can often find in raw food.