Thomas Schultz on Wikimedia Commons
The gut microbiome can hit fast forward on Alzheimer's disease progression
Male — but not female — mice had reduced amyloid beta plaques in the brain after antibiotic treatment
For approximately every human cell in our bodies, there is a tiny microorganism living inside us. On our skin, in our lungs, mouth, gut and more. Collectively, these microscopic bacteria, viruses, and fungi are called the microbiome. These organisms sometimes merely coexist, sometimes harm their human hosts, and sometimes provide benefits.
The community of bacteria that live in the gastrointestinal tract — the gut microbiome — is generally harmless, but, because these bacteria affect the activity of the body's immune system, they can influence a wide range of diseases, even in distant organs like the brain.
Recently these microorganisms have become closely associated with diseases like cancer, diabetes, and Parkinson's. In a study published in the Journal of Experimental Medicine earlier this year, microbiologist Hemraj Dodiya of the University of Chicago and colleagues sought to determine whether bacteria in the gut could influence the progression of Alzheimer's disease.
Dementia affects 50 million people worldwide. Alzheimer's disease is one of the most common forms of dementia; it is characterized by memory loss, confusion, and other cognitive symptoms that gradually progress into near-total dependence and immobility. About one-third of people that live until age 90 will develop a form of dementia. There is no cure. Drugs can ameliorate some symptoms but cannot stop the progression of the disease.
Although we don't understand exactly how Alzheimer's causes these symptoms, the disease is partially characterized by a buildup of protein clumps in the brain, called amyloid-beta plaques, which appear along with cognitive decline. Though amyloid beta accumulation characterizes the disease, much remains unknown about how these plaques wreak so much havoc in the brain.
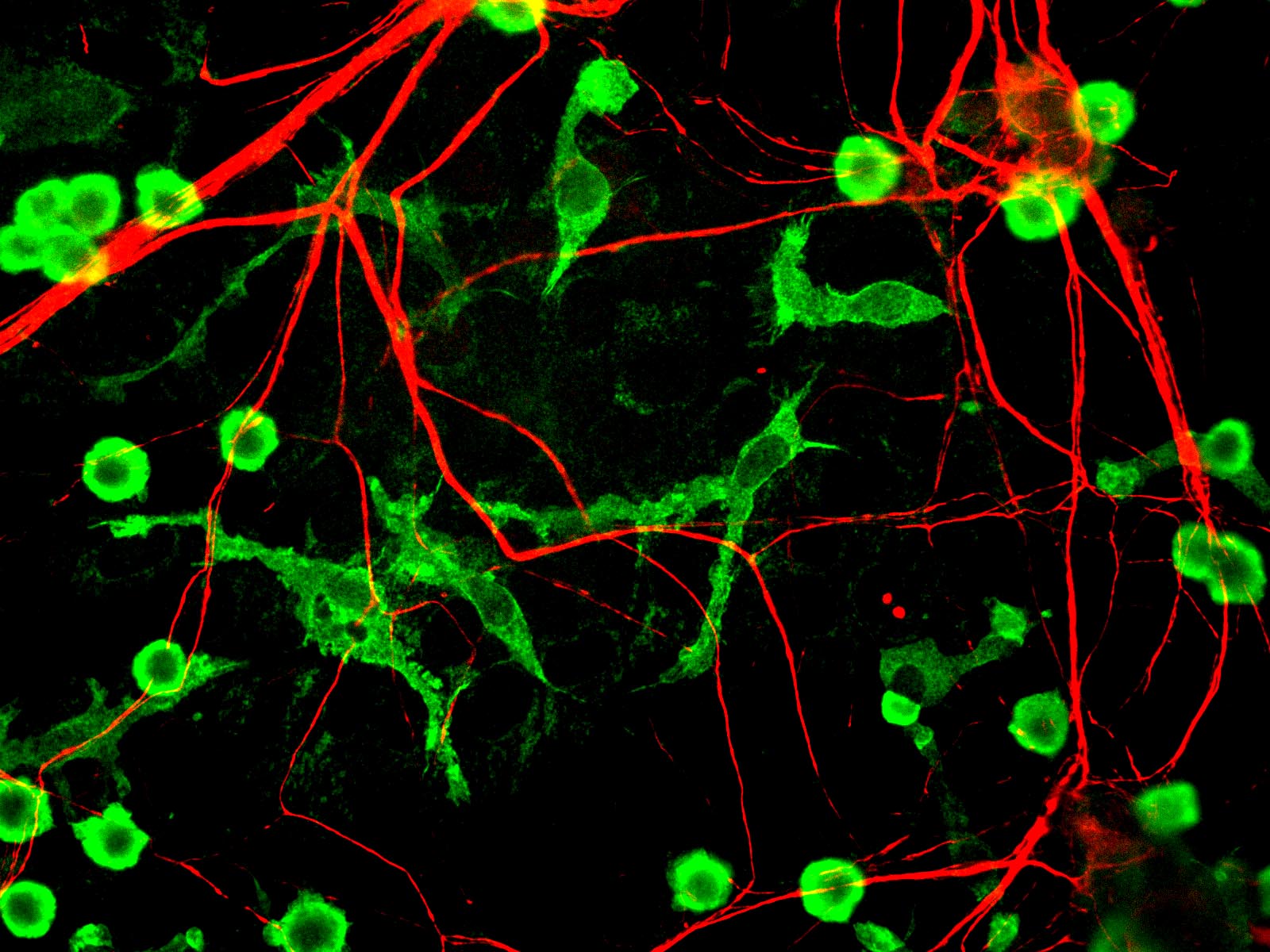
Microglia (green) and neurons (red)
GerryShaw on Wikimedia Commons
Normally, the immune system plays an important role in removing amyloid beta, but scientists believe the immune system might also perpetuate and accelerate Alzheimer's. Microglia, immune cells that live in the brain, look for harmful substances and danger signals. When they run into amyloid beta plaques, microglia shift to a pro-inflammatory state. When this happens, the microglia release inflammatory chemicals that, at high concentrations, may actually make Alzheimer's symptoms worse and contribute to brain cell death. Whatever their role may be, this inflammatory state reflects an impaired capacity to maintain healthy tissue, not only in Alzheimer's disease, but in other neurodegenerative disorders such as Huntington's and Parkinson's diseases as well.
How do microglia morph from helpful amyloid beta-eating machines into dangerous purveyors of excessive inflammation that can end up killing neurons? While a number of factors are surely at play, changes in gut bacteria might be a key player in triggering or sustaining inflammation-inducing microglia. In their study, Dodiya and his colleagues found evidence that bacteria can influence Alzheimer's symptoms in male mice.
They tested the effects of microbes in the gut on amyloid beta and microglia in the brain using a line of transgenic mice developed specifically to have extra amyloid precursor proteins. First, the researchers administered antibiotics to the mice, which altered their gut microbial communities. When they looked at brain tissue from those mice, they found that their microglia had stayed in check and these mice had fewer amyloid beta plaques. To confirm that the change in microbial communities could contribute to these effects, they then reintroduced microbes by transplanting fecal matter from healthy mice back into another group of mice that had been treated with antibiotics. Reintroducing the microbes partially restored the amyloid beta plaques and increased markers for inflammation-causing microglia. And although the researchers tested both male and female mice, they only found these changes in male mice.
Rama on Wikimedia Commons
For unknown reasons, women are more likely to be affected by Alzheimer's than men. The researchers considered this fact and accordingly, tested both male and female mice. The microbiome's effects on plaques and microglia only applied to male mice: amyloid beta and microglial levels did not change in response to antibiotics in female mice. Researchers often use male mice, based on historical precedent and bias, but this doesn't reflect the reality of how our bodies function. Diseases are often more common in one sex; Alzheimer's appears more frequently in females, but disorders like Parkinson's appear more frequently in males. On top of that, the microbiome effects can also be sex-specific, regulating sex hormone levels and autoimmune disease fate. This is not the first study to compare how the microbiome has different roles in different sexes, but it highlights the importance of considering sex when thinking about therapeutic strategies for Alzheimer's and other diseases.
Although we're not likely to start giving long-term antibiotics to treat Alzheimer's disease, the finding that antibiotics reduced Alzheimer's symptoms in male mice can now be harnessed to better understand timing and influence of gut bacteria in Alzheimer's disease. Is there a window of time in which bacterial function is critical? In the future, will we be able to seed our guts with specific bacteria to reduce Alzheimer's symptoms? More studies are needed to understand these processes and extend the lessons learned in mouse models to human disease.



The gut microbiome (GMB) affects the immune system by direct contact with immune cell receptors, and by chemical signaling via the molecular products of microbial metabolism of dietary fiber. Most researchers now consider the GMB more like an organ than a symbiont, an inherent part of us like mitochondria, so it’s a bit odd to refer to it as either harmless or harmful. Despite our knowledge of pathogenic microbes that can cause disease, many of these can be docile members of the GMB in healthy, asymptomatic individuals. Our historical perception of the GMB was limited to low resolution snapshots of diversity, which has been changing as DNA sequencing costs continue to go down and with the advent of computational techniques that track patterns in large & longitudinal datasets. A good example of the evolving ecological view of the GMB can be found here. On that note, I have a feeling this type of longitudinal sampling and analysis is the key to unlocking GMB-Alzheimers interplay in humans…which is to say, we won’t know anytime soon, but at least we are finally grasping the tools!