Haley Lawrence on Unsplash
Microglia may be the missing clue to solve the opioid epidemic
These tiny cells have the machinery to sense opioids, and targeting them may relieve opioid addiction
Chronic pain—pain that lasts or reoccurs for more than three months—doesn't often make headlines. But it's a public health epidemic that affects 20 percent of people worldwide. To make things worse, efforts to manage and treat chronic pain have been complicated by the opioid epidemic. In the United States alone, more than 11.4 million people misuse opioid drugs every year. To try to control this crisis, American legislators have begun to closely regulate prescriptions and limiting access for chronic pain patients—those who could benefit from these drugs the most. But now, neuroscientists have discovered a new therapeutic target for managing pain: microglia.
Pain, as an acute sensation, serves as a warning to help your body prevent injury or avoid further harm. When, for example, you stub your toe, nerves in your foot send an electric signal that travels through the neurons of your spinal cord before finally arriving in your brain, where many regions including the brainstem integrate the message into your perception of pain, both physical and emotional. Your brain then sends an electric signal back down to your toe. These ascending and descending pain pathways can either either amplify or reduce your perception of pain, which researchers back in the 1970s termed the "gate-control theory of pain." Essentially, the message from your stubbed toe is forced to go through a series of checkpoints—or gates—which will either open or shut to control the intensity of pain you perceive. When pain becomes chronic, this signaling and the gate controls go haywire. Nerves become hyper-sensitive, firing off messages to the brain even in the absence of an injury.
An emergency naloxone kit.
James Heilman on Wikimedia Commons
Traditionally, researchers trying to untangle pain pathways have focused on neuronal connections. Opioids are particularly effective at relieving pain because neurons in the brain and spinal cord contain specialized receptors, called Mu opioid receptors (MOR). Opiodergic drugs, like morphine or oxycodone, bind to these receptors, overriding the pain signals between neurons and creating an analgesic affect. Drugs like Naloxone, used in cases of overdose, basically just unbind this connection, reversing the effects of the drug. But although it may save the life of someone overdosing, Naloxone alone doesn't solve the problem of opioids' dangerous side effects and dependence.
Fortunately, new research suggests another key player in how pain works: microglia. Microglia are the smallest of the glial cells found in the brain and spinal cord, and act as the primary system of defense in the central nervous system. They are essentially first-responders in cases of injury, helping to guide the immune response. Microglia are developed in the brain in utero, and are active in its overall maintenance. And recently, researchers from France have found that microglia express, or produce, opioid receptors. These receptors are located on the outside of the cell, or its membrane. When an opioidergic drug binds to this receptor, it will change its configuration, and ultimately the action of the cell. This means that opioidergic drugs, like methodone, don't only act on neurons—it turns out they have a direct impact on microglia, too.
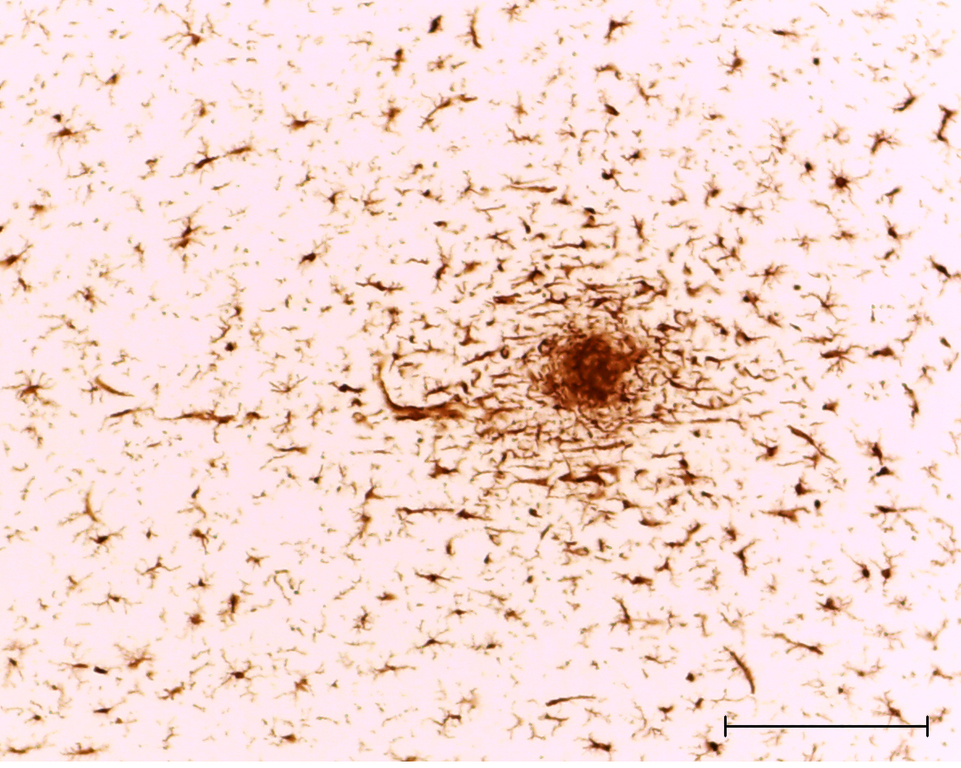
Microglia migrate towards damage in neuronal cells. Scale bar is 50 microns.
Mary Antipova on Wikimedia Commons
While microglia have previously been shown to interact with neurons, their exact roles are still not entirely understood. Previous studies investigating the role of microglia had not been able to definitely prove that microglia interacted with opioidergic drugs. However, these studies were done on cultured microglia grown in a lab dish. Scientists know that these kinds of cultured cells can have very different gene expression than those found in vivo, meaning in a living organism. And scientists who did try to find MOR expression in microglia in vivo had contradictory findings; they couldn't conclude if the drug interaction was with microglia directly, via the involvement of other cell-types.
But finally, an interdisciplinary team from France decided earlier this year to use a handful of large datasets from other researchers who have compiled genomic data specifically for this kind of analysis. They used transcriptomic analysis—looking at the complete set of RNA transcripts produced by DNA—on both human and animal subjects. Their goal was to isolate the specific gene responsible for expressing the MOR in microglia, Oprm1/OPRM1. Then, the researchers genetically designed mice with fluorescent cells that light up if they have this gene, allowing the scientists to visually see the MOR protein in microglia. The researchers ultimately successfully found microglia with MOR expression in both the brain and spinal cord. By showing that these microglia carry the genetic code for opioid receptors, the study supports the hypothesis that microglia are directly involved in opioid tolerance or addiction—contradicting previous research.
Of course, this study was not without its limitations. First, most pain research has been done on (cis) male subjects. (Sadly, this is not unique to pain research—almost all neuroscience research involving non-humans has a strong 'sex-bias.') While the NIH has recently mandated that researchers justify their research methods including choosing the sex of their subjects, by using already existing datasets, this study lacked data from female subjects. Secondly, the human data was limited: Most of it came from the brain, not the spinal cord. Due to the overall volume of data, the researchers could only study a few brain areas, so not all the regions involved in addiction or pain were thoroughly investigated. Finally, there is still some controversy on the differences between phenotypes of microglia—some researchers believe that there may be different kinds of microglia with different jobs, complicating contradictory results even further.
But overall, these insights should inspire researchers, clinicians, and chronic pain patients alike to consider new possibilities. There are already a few drugs available which target microglia, that could now be considered for pharmaceutical interventions for pain reversal or prevention. In addition to pain relief, targeting microglia might also be a way to prevent opioid tolerance, or explain opioid-induced hyperalgesia, when the exposure to opioids actually makes the sensation of pain worse. Future combined drug strategies might allow patients to have the pain-relieving effects of opioids without the unwanted or dangerous side-effects or addiction.
Since the study's publication, other researchers and clinicians, such as Dr. Vivianne Tawfik’s team at Stanford, have called for more nuanced pain research. This kind of research should include not only variables like the sex and age of patients, but also supports the need for more foundational pre-clinical research. Understanding microglia, which we still don't know enough about, may help bridge the gap between the bench and the bedside— and may ultimately be the missing clue in solving the opioid epidemic.


Nice article, very well written!
The role of microglia in pain processing is so poorly understood! This article from the French team underscores the extensive problem of conflicting results in pain research.
The results made me think of recent work demonstrating that opioid signaling through pain receptors (neurons) and not microglia in the periphery is required for morphine tolerance (Corder et al., Nat Medicine 2017). At the same time, these authors did not find opioid receptor expression in microglia, which is at odds with the French report. So the jury is still out on the role of opioid receptors in microglia and the contribution of microglia to pain processing.
While microglia may contribute to opioid processing, we’re probably not going to be able to ignore neurons when thinking about targets for addiction, especially as the circuitry for addiction operates at the level of the brain - likely parallel to or in concert with the pain processing pathways.