
Photo by Gabriel Crismariu on Unsplash
Healthy and cancerous pancreatic cells fit together in a dangerous puzzle
Salk scientists are piecing together a picture of one of the deadliest cancers
Dannielle Engle was in college when her family received some news that would change her direction in life forever: her father was diagnosed with late-stage pancreatic cancer. He had few treatment options, and none of them were very promising.
Pancreatic cancer is one of the deadliest cancers because is often symptomless, leading to late diagnosis only after metastases have spread throughout the body. The tumor is also often surrounded by an impenetrable shell of fibers, proteins and immune cells, which makes it difficult to target and treat.
The Salk Institute for Biological Studies, where Engle is a faculty member, is one of only seven National Cancer Institute Basic Laboratory Cancer Centers in the United States. Researchers there are focusing their efforts on finding a cure for some of the deadliest cancers, including pancreatic cancer. Their studies, which have appeared in numerous journals and led to clinical trials using novel approaches, are coming at pancreatic cancer from different angles, reflecting the idea that a problem as complex as pancreatic cancer requires a multifaceted solution.
“Pancreatic cancer is incredibly aggressive, but that's not necessarily because it moves very fast,” says Engle in a recent podcast interview. “It is very aggressive because there aren't any early warning signs. All the symptoms are very vague and by the time you start feeling those symptoms, it is often already too late.”
Given her personal history, it’s perhaps no surprise that Engle is focusing on the developmental stages of pancreatic cancer. In particular, her lab is interested in the transition between inflammation of the pancreas, known as pancreatitis, and the early stages of pancreatic cancer development. Having the ability to detect pancreatic cancer early on could lead to an increased survival rate and better prognosis for patients.
Pancreatitis can transition into pancreatic cancer, just like pre-diabetes can advance into diabetes. Currently, the only FDA-approved biomarker, a signal used to detect a disease, for pancreatic cancer is CA19-9, a complex sugar structure that coats many proteins, but which can also be present in pancreatitis, making it difficult to differentiate between the two diseases.
Recently, Engle found that CA19-9 may actually cause the progression of pancreatitis to pancreatic cancer. Her research suggests that blocking CA19-9 could be used therapeutically to prevent disease advancement.
Engle is also studying pancreatic cancer by modeling the tumor growth in the lab using organoids. Organoids are tiny, three-dimensional clusters of cells, derived from patient skin biopsies, that mimic organs such as the pancreas. Engle is building a repository of pancreatic organoids, derived from both healthy and cancerous tissue, to study the differences between the two tissue types. She hopes that knowledge gained from these studies could lead to better tools for diagnosing cancer.
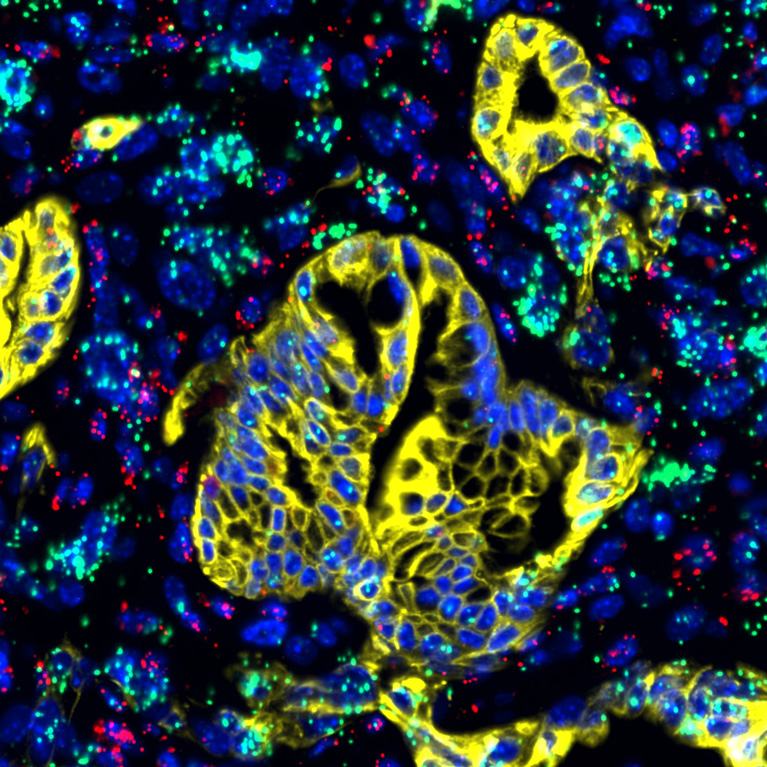
A marker (called LIF) for healthy pancreatic stellate cells seen here in green, alongside immune cells (purple) and pancreatic cancer cells (yellow)
Salk Institute
“My hope is to make scientific discoveries that make a difference to patients and families,” says Engle.
Another professor in Salk’s Cancer Center, Tony Hunter, is known for his 1979 discovery of a genetic switch that, when flipped, can turn cancer on or off. Malfunctions of the switch, called tyrosine phosphorylation, often lead to cancer.
This fundamental knowledge about tyrosine phosphorylation led to the development of drugs that can target this switch, such as the anti-leukemia drug Gleevec.
Most recently, Hunter has focused his research efforts on studying pancreatic cells called stellate cells. These cells can become inflamed and begin communicating with tumor cells in ways that promote pancreatic cancer growth, in a very snake-eating-its-own-tail way.
“It's sort of a vicious cycle, if you like, of both cells maintaining each other,” explained Hunter in a recent podcast interview. “We generated a catalog of all the proteins that the stellate cells make, using a technique known as mass spectrometry, to identify these proteins involved in this communication.”
His team found that pancreatic tumors have high levels of a protein called leukemia inhibitory factor (LIF). Further analyses revealed that LIF was actually promoting tumor growth.
“We found out was that we could detect LIF in the serum of both mice and importantly in the serum of pancreatic cancer patients,” says Hunter. So, it could potentially be used as a biomarker for response to therapy in people.” A biotechnology company called Northern Biologics has already developed an antibody that targets LIF, and phase 1 clinical trials for the drug began in August of 2018.
Photo by Allie Smith on Unsplash
In addition to Hunter and Engle, other Salk researchers have made groundbreaking strides in pancreatic cancer. Professor Ronald Evans discovered that a chemically modified form of vitamin D makes tumor cells vulnerable to chemotherapy and more sensitive to the body’s immune system. Evans’ team launched a clinical partnership to test this drug in pancreatic cancer patients. Professor Geoffrey Wahl’s lab is creating a strategy to weaken the impenetrable shell around the tumor, thereby disrupting the tumor’s “control center” to make it more sensitive to therapeutic intervention. And Professor Susan Kaech is bringing yet another approach to understanding pancreatic cancer by focusing on deciphering how T cell activity, cells critical for maintaining long-term immunity, is suppressed by tumors. Her aim is to create better cancer therapies using the body's own immune system.
To learn more about Salk researchers’ life-changing work in pancreatic cancer and other areas, tune in to the Where Cures Begin podcast on Salk.edu/podcast, iTunes or anywhere your listen to podcasts. You can read more about Salk’s pancreatic breakthroughs in a recent feature magazine article “Stopping a Killer.”

