
How shaving mice led to an accidental discovery about our sense of touch
We're gonna need more shaving cream
The average human lifespan is around 80 years, but the cells in our bodies live, die, and grow again along much shorter lifespans. Cells turnover at different rates and for different reasons. Consider the skin: it’s constantly exposed to hazards including both physical abrasions, and harmful UV rays that cause mutations.
Because of these hazards, our skin cells replace themselves about every month to make sure that our skin stays strong and mutation free. This is starkly different from neural tissue, the infrastructure for sending electrical signals throughout the body. Some neurons never turnover, because many neurons are large, branched cells that are very difficult to replace without disrupting their important functions.
Then there’s the Merkel cell, which helps detect the sense of touch. We have several types of cells in our skin that detect different types of touch, like pressure, vibrations, or temperature. Merkel cells are the closest to the skin's surface, allowing us to detect the gentlest sensations. If you close your eyes, you can easily detect the subtle difference between a hard granite or a relatively soft marble countertop, and you have the Merkel cell to thank.
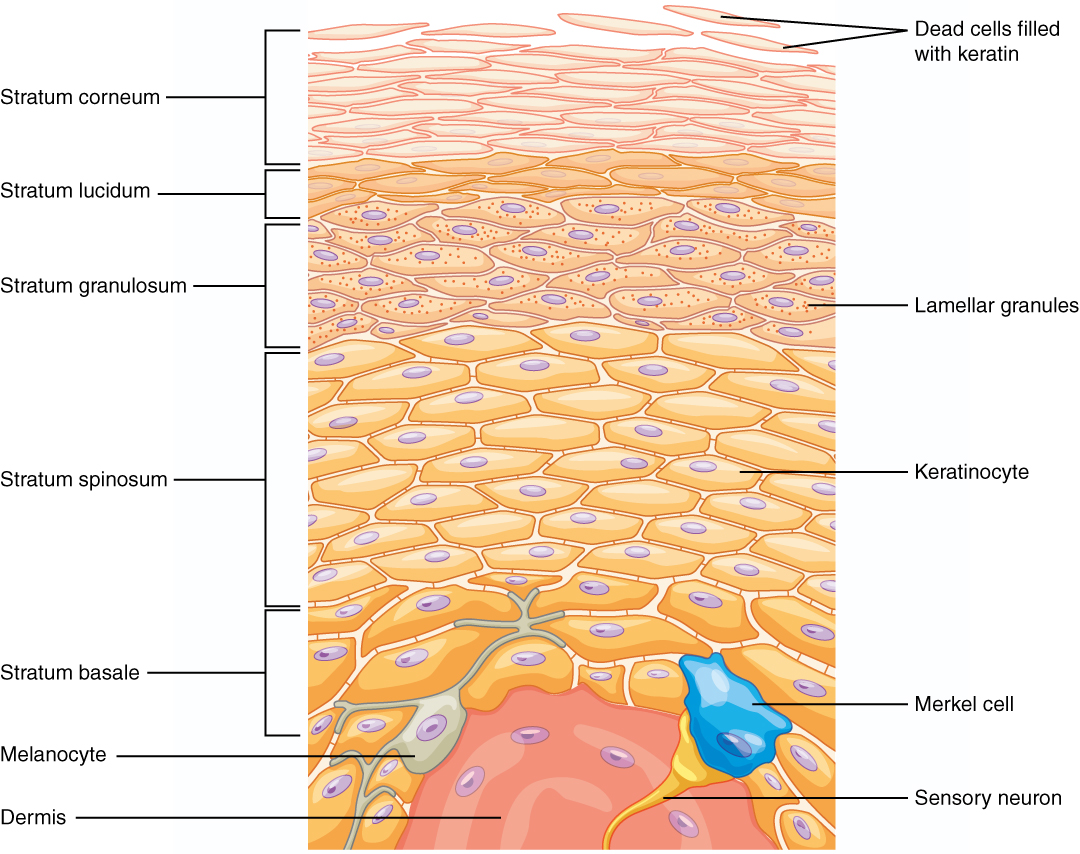
A diagram of skin layers, showing Merkel cells at the bottom.
But the Merkel cell presents a conundrum when we think about lifespan. It’s a skin cell, so we would expect it to turnover frequently; however, it functions like a neuron, forming synapses with sensory neurons, so we would also expect a slow turnover rate. For this reason, scientists still debate turnover rate of Merkel cells.
Why do we care about Merkel cell turnover? For one, we want to understand more about how Merkel cells repair after injury, a question that matters enormously to burn victims and the elderly, who have a diminished sense of touch. Understanding more about how Merkel cells develop may result in therapies to help restore the sense of touch in these individuals. But cell turnover matters most for understanding cancer, which is often driven by mutations in genes that regulate turnover. A rare but deadly cancer called Merkel Cell Carcinoma is caused by skin cells that appear to change in part into Merkel cells.
Many scientists believe that Merkel cells turnover frequently as most skin cells do; however, our lab thought this didn’t make sense, so we set out to test if this was true. To test this hypothesis, we used a technique called lineage tracing, a method created by developmental biologists to ask what happens to a cell over time.
To make lineage tracing work, scientists take genes that are expressed in specific cells and reprogram them to produce a marker that we can see. This allows us to track a cell like GPS tracks your location. We used this technique to put a marker on all of the Merkel cells in a mouse, then we came back nine months later to see what happened to them. Surprisingly, we saw that all the Merkel cells still had the marker. The Merkel cells did not turnover at all.
But we weren’t finished yet. We knew that other scientists would have a hard time believing us, so we wanted to bolster our hypothesis with a different model, and perhaps simply use a microscope to look at Merkel cells in living mice. Luckily, we had a strain of transgenic mice, animals with foreign genes introduced, which produce a fluorescent protein in their Merkel cells. We took these mice, shaved off their hair to get as close to the skin as possible, then placed them directly under a microscope. To our surprise, through all the layers of skin, we saw clusters of bright green Merkel cells – the experiment worked!
But we still wanted to know whether Merkel cell numbers changed overtime, so we went to the CVS down the street and bought some Barbasol. Every week, we brought the same group of mice to my lab bench, covered their abdomens with shaving cream, and shaved their bellies with a disposable razor. Then we carried them to the microscope and measured the number of Merkel cells in their skin. After a few weeks, we noticed a few new Merkel cells arising in different parts of the skin. We were seeing turnover.
Get ready for a shave.
We were baffled. In our first experiments, we measured no turnover of Merkel cells. It seemed that Merkel cells lived forever. But in this new experiment, we were seeing new Merkel cells produced every few weeks. The difference couldn’t be due to error, so what else could it be? We laid out the experiments and wondered what had caused the discrepancy. Only one difference stood out: in the second experiment we were shaving the mice.
Could shaving cause Merkel cell turnover? We think it could. When you shave, you don’t just cut the hair, you scrape the top layers of skin, creating a skin abrasion. Human skin is thick, so we may not notice. Mouse skin, however, is only a few cell layers deep, so removing a layer of cells could cause a wound response. We believe that wound response is causing Merkel cell turnover.
From these experiments, we concluded that Merkel cells almost never turnover. Under normal conditions, they rest in your skin and help you detect physical stimuli around you, and don’t usually turnover, because this would disrupt your sense of touch. However, since they sit in an area of the body that receives frequent skin injury, it makes sense that a repair mechanism exists. It seems that injury increases Merkel cell turnover.
We still have more to discover about what causes the production of Merkel cells. Some scientists don't agree with us. Their experiments show that Merkel cells turnover quite frequently, so we will first need to repeat a few experiments to determine if an error was made. Still, our experiments tell us that skin shaving seems to cause Merkel cell production, but what's happening under the skin? After shaving, a wound response causes a release of many molecules to promote regrowth of the tissue, and we would like to look at these specific molecules to see which ones promote Merkel cell growth. Could these same molecules cause progression of Merkel cell carcinoma?
We'll need more shaving cream to find out.




I am curious about the correlation between the two mouse models used. It does seem possible that the shaving is inducing the Merkel Cell turnover in the transgenic mice, but would the same effect be visible if you regularly shaved the initial lineage-tracing mice? I.e., when you take the tracing mice down, those who had been shaved regularly would have more non-tagged Merkel Cells since there would be new generations of cells in response to the skin manipulation. In the same vein, could the transgenic line be crossed into a hairless background, and then you could see if the fluorescent cells change without any shaving at all? If both are true, then I would completely agree that shaving is the culprit. But otherwise, how much of this effect could be based more on the mouse used than the skin manipulation?