
Patrick Hendry via Unsplash
CO2 sucked out of the atmosphere can be reused for new chemical processes
It can be converted into methanol, then used in resins and polymers
Global warming is the biggest challenge we have ever faced.
Since the first industrial revolution, the amount of carbon dioxide (CO2) — a powerful greenhouse gas — in the atmosphere has increased by almost 50%, which is causing a steep rise in global temperature. Controlling its increase and keeping temperature rise below 1.5 degrees is necessary to avoid the most catastrophic effects of climate change. However, that likely requires stopping new emissions immediately and reducing the excess CO2 that is already in the atmosphere.
Scientists are developing technologies that can literally suck CO2 from the air with huge fans. This technique, called direct air capture (DAC), uses either adsorption — where CO2 sticks to a surface — or chemical reactions which capture and use CO2 to create something else.
DAC is implemented in pilot plants by several companies in Europe and North America, and on an industrial scale by a Swiss company called Climeworks. They plan to suck one percent of the global CO2 emission in 2025. For this task, they will need to install around 750,000 DAC units.
After removing CO2 from the air, DAC plants have to get rid of it. One way to do that is to pump it underground, where it essentially turns into rock. They can also sell the CO2 for use in greenhouses (CO2 is a potent fertilizer) and carbonated drinks, or fuel, polymers, and concrete.
Coming up with new ways to use commercialize extracted CO2, once it's sucked out of the air, can inspire new technology that reduces atmospheric CO2.
Recently, scientists from the University of Southern California have developed a simple method to turn CO2 into methanol. The conversion happens without complicated purification or separation steps.

Gas combusting in a flare
In a more traditional method, the separation of the captured CO2 requires temperatures higher than 700°C. In this novel technique, potassium hydroxide, a base, captures CO2 and forms a compound called potassium formate. At this point, adding hydrogen should, theoretically, produce methanol. However, it turned out to be not that easy. This step represented the main challenge that the researchers had to face. As they write, in baldly scientific style, "further hydrogenation [addition of hydrogen] of the formate...was found to be ineffective."
Going back to the literature, they tried a new approach, which they hoped would lead to producing methanol but through a different route. They dunked the whole reaction in alcohol, added a catalyst to speed it up, and increased the temperature, obtaining the total conversion of CO2 to methanol in 20 hours.
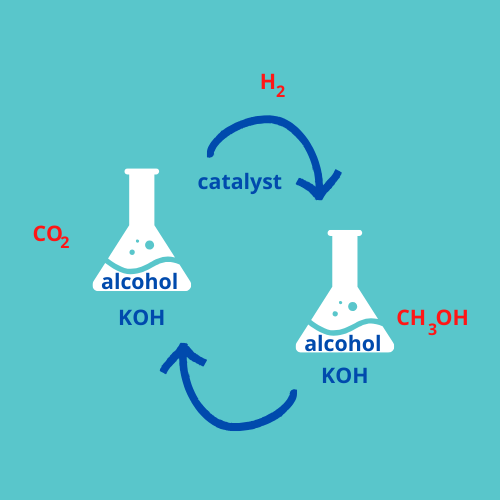
Simple "one-pot" conversion of carbon dioxide (CO2) into methanol (CH3OH)
Rosaria Cercola
This method still requires some adjustments. The reaction is cyclical — you end up with the same material in the pot, plus the methanol, at the end of the reaction as you had in the beginning. Different components wander off and turn into other chemicals, but are regenerated at the end in the same proportion they were in at the beginning. But the base does gets consumed. If the researchers can overcome this issue, this technique can be easily scaled up and added to existing plants.
But, there's more. For it to be energetically efficient, the production of hydrogen must be derived from splitting water with renewable sources of energy (such as from solar power). This method is not widely available yet, but it is being tested in a series of HYDROSOL projects.
Right now, most of the hydrogen used in the chemical industry comes from fossil fuels. The steam reforming method — cheap and common but energetically demanding — heats up methane gas and water at very high temperature (700-1100 °C) to produce carbon monoxide and hydrogen. With these plants popping up more and more, we could see large-scale sustainable CO2 to methanol conversion in a few years.
But then what will do we do with all of that methanol?
We can either use it as it is, as fuel or antifreeze, or as a reactant to produce more chemicals. The first case is understandably not the solution to our CO2 problem: as a fuel, methanol burns and produces new CO2 that goes back into the atmosphere. This process, described as it is, is a carbon-neutral process, and not carbon-negative as we need. Subtracting CO2 from the atmosphere is not enough if global emissions do not decrease.
On the other end, methanol can be used to produce solvents, resins, low weight polymers, long-lasting materials that don't liberate CO2 immediately. However, for it to be carbon negative, it is necessary to consider the balance between the stored carbon and the one produced during the synthesis of these materials.
More investments in this kind of research are necessary to reduce the production of CO2 altogether. This topic is delicate and involves scientists, policymakers, and the choices consumers make along the way: The cars we buy (or rather don't), the flights we take (or rather don't), the electricity source we choose for our own houses. But by and large, the people that we elect to lead us in these decisions.



A lot can be done to decrease the emissions of CO2 but it is also interesting how somehow, with the technique you mentioned in the article, the CO2 built up for years can be reduced at some point in time.
Regarding the conversion of CO2 into methanol, I am intrigued by how methanol was separated from the other alcohol used.
Lastly, I was very compelled by “pump it underground, where it essentially turns into rock.” CO2 is a gas, so how was it converted into solid stone? For anybody who also found this interesting, the study indicates CO2 (and H2S) is dissolved in water (basically like making a diluted sparkling water) and injected underground which contains calcium, iron, and magnesium enriched volcanic rocks. It turns out this process can generate calcium, iron, and magnesium carbonates. Which is very cool! If you're interested in learning more, I'd recommend this.