Illustration by Allan Lasser

A version of this story originally appeared on Reclamation
We still don't really know how Zika gets into the brain
For this scientist, the race to understand one of the world's most mysterious diseases is personal.
In February of 2016, the Zika virus was marching north from Brazil through Central America, disproportionately affecting the poor. Over 70 years since its initial discovery in the Zika forest in Uganda, the virus suddenly went from minimally impacting human health to dominating international headlines because of its devastating effects on fetuses.
Unlike close relatives like West Nile, Yellow Fever, and Dengue, the Zika virus found a way to cross the placenta from mother to fetus. This lead to thousands of cases of infants being born with small brains, a condition known as microcephaly (micro meaning small, and ceph meaning head). Heartbreaking images of these babies began to fill our TV and computer screens, showing the world just a fraction of the immeasurable damage this virus was doing to some of the most vulnerable people on the planet.

Around this time, my boss Dr. Kevin Eggan at Harvard University came to me with a proposal. He suggested that I use the tools we have at our disposal as neuroscientists to study the effects of the Zika virus on the brain. For me, this was more than just a chance to investigate a strange disease. It was an opportunity to connect with my Latin roots.
I’m not sure why, but I was never able to learn how to speak Spanish. Unlike many of my fellow non-bilingual Americans, I have no excuse for my ignorance. My mother was born and raised in El Salvador, and despite her many attempts, I could never quite grasp her native language. As a result, I am silent at large family gatherings, unable to understand the heroic stories of my grandmother, who raised three girls on her own during the Salvadoran Civil War. Though Mestizo blood nourishes the fingers that type these words, I often feel deeply disconnected from the place my ancestors called home, and where much of my family still lives.
When my boss first approached me, El Salvador already had over 6,000 suspected cases of Zika. I saw this study as a chance to learn, contribute, and most importantly, fight for my family 2,000 miles away. We quickly assembled a team at Harvard and the Novartis Institutes for Biomedical Research and got to work.
Understanding an emerging threat
Before Zika became a household name, a group of scientists in France had already taken some of the first important steps towards understanding this mystifying virus. By infecting skin, lung, and kidney cells on a plastic dish, they started investigating how Zika is able to enter those types of cells. In October 2015, the paper describing their work was published and quickly became a must-read for Zika scientists everywhere. In fact, my colleagues and I refer to this paper as the Zikapedia.
One of the important things that these scientists discovered was that a protein that some of our cells produce called AXL plays a critical role in Zika infection. (Disclaimer: To our knowledge, this protein was not named after Guns N’ Roses lead singer Axl Rose, though we hope we’re wrong).
The Zikapedia group identified the relationship between AXL and Zika by performing simple, yet informative experiments. They added different compounds to cells before and during infection, and found that when drugs that blocked or eliminated AXL were used, the virus was unable to enter and kill those cells. Thus, they concluded that AXL helps Zika invade cells.
At this point, it’s important to mention that what’s true for one type of cell may not be for another. When Zikapedia was first published, most researchers didn’t suspect that Zika could infect the brain cells of fetuses, since this is not a common feature of viruses transmitted by mosquitoes. So, while Zikapedia was helpful in finding paths the virus can use to enter skin and lung cells, we were left not knowing the answer to one critically important question—how does the Zika virus get into brain cells?
Turning off the brain cell assembly line
To answer this question, several research teams first determined that the virus prefers to infect a type of brain cell called neural progenitor cells. Early in the development of a fetus, these progenitors multiply in number and populate the brain with both neurons – the brain cells that communicate with each other through electrical and chemical signals—and glia cells—the brain cells that support and protect neurons.

From this piece of information, it became obvious how Zika was causing microcephaly. By infecting and killing fetal progenitors, Zika shuts down the factory that produces neurons and glia. As a result, the fetuses develop fewer brain cells and smaller brains.
Influenced by this early work, a group in California led by Dr. Arnold Kriegstein looked at different cell types in the brain and found that the cells that are susceptible to Zika infection are coated with our old friend, the protein called AXL. Based on this correlation, these researchers hypothesized that the Zika virus was entering the progenitors and other brain cells with the help of AXL. If this were true, it would mean that we could prevent or at least slow down Zika infections by interfering with the AXL protein, just like researchers who worked on the Zikapedia paper did with non-brain cells.
A few months after publishing this work, the Kreigstein group showed that blocking AXL effectively prevents the virus from infecting glia cells that reside in the brain. The AXL-Zika argument was getting stronger and stronger. At this point, my team was convinced that AXL was just as important in brain progenitor cells.
A disappointing, yet informative result
Fueled by these findings, my team created human progenitor cells that were missing all traces of AXL. We cut AXL out of these cells using the CRISPR gene editing system, which essentially acts like microscopic scissors for DNA.
After removing AXL from the equation and checking to make sure that the loss of this protein did not cause any major health problems for these cells, we asked a simple question: “can the Zika virus still get in?” If the answer was no, it would suggest that Zika needs AXL in order to penetrate progenitor cells. If the answer was yes, it would imply that either AXL is not important at all or, more likely, that there are other routes for viral entry into this type of brain cell.
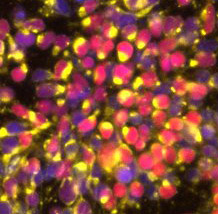
Much to our surprise and disappointment, when we added the virus to both types of cells, the cells without AXL showed the same levels of infection and died at the same rates as normal cells with AXL. The absence of AXL from the outer coat of progenitor cells did not protect them from Zika infection, a finding independently corroborated by another group using a slightly different method. Studies in mice missing AXL also showed this result, which is important because cells in a dish don’t always behave the same way as cells in a living animal.
Even though we didn’t get the results that we had hoped for, we still revealed something important. Now we know that AXL is not the only “door” that Zika can use to enter progenitor cells. We believe that progenitors produce other access points for the virus, meaning that even though we sealed off the AXL entrance, we may have unknowingly left another door open to Zika.
Hopeful uncertainty
We may not have won this battle, but I am optimistic that we, or someone else, will. My team is attempting to answer some of the remaining questions surrounding this enigmatic virus through a series of follow-up experiments. If we can figure out how the virus gets into progenitor cells, perhaps someday, someone will figure out a way to safely disrupt this process in order to prevent future cases of Zika-related brain deformities.
I spent the past 12 months of my life trying to understand a virus that connects me to the family members I've been struggling to understand for 31 years. Though we don’t know much, I do know that unlike me, the spread of the Zika virus can’t be hampered by language barriers. If we don’t try to stop it, or at least understand it, we will all surely find ourselves in the same situation as my beloved El Salvador - in the crosshairs of a vicious infectious disease with few weapons at our disposal. I don’t know if we’ll succeed, but we must keep trying.
Featured Article
- Wells, Michael F. & Salick, Max R. et al. Genetic Ablation of AXL Does Not Protect Human Neural Progenitor Cells and Cerebral Organoids from Zika Virus Infection. Cell Stem Cell, Volume 19, Issue 6, 703 - 708. http://dx.doi.org/10.1016/j.stem.2016.11.011.
