
Adapted from the Smithsonian Institution via Wikimedia Commons.
Meet Gerty Cori, the Nobel-winning biochemist who uncovered how the body stores and consumes sugars
Cori's work determined glycogen storage "disease" had several subtypes, each with a unique molecular cause
In 1947, Gerty Cori traveled to Sweden with her husband, Carl, to jointly accept the Nobel Prize in Medicine and Physiology. It was an undoubtedly exciting moment. The Coris had received the Prize for their successful test tube synthesis of glycogen. From their discoveries, scientists now knew exactly how glycogen was processed in the body to create glucose – a once mysterious metabolic process that had confounded researchers for decades.
Gerty Cori became the third woman, and the first American woman, to win a Nobel Prize in science. The summer before the prize ceremony, however, Gerty had developed myelosclerosis – an incurable and ultimately fatal disease. To Gerty, however, the diagnosis only served as further fuel for the scientific questions still lingering in her mind.
And so, after the ceremony, she returned to the laboratory, eager to resume her experiments. Gerty was intensely curious about how glycogen, when mutated, caused disease. This group of rare diseases, called glycogen storage diseases, manifested through an enlarged liver, weak muscles, and growth obstruction. What was the crux of the problem that caused the body’s symptoms?
Gerty Theresa Cori (then Radnitz) was born in Prague in 1896. The daughter of a sugar refinery chemist, Gerty was a precocious child with a clear interest in the sciences. She entered medical school at the German University of Prague in 1914, where she met another young scientist – Carl Cori, who was smitten by her intelligence, charm, sense of humor, and love of the outdoors. In 1920, Gerty received her medical doctorate, married Carl, and published their first joint research paper together. As a postdoctoral researcher, Gerty began her research career by studying pediatric diseases at the Karolinen Children’s Hospital in Vienna.

The Coris working in the lab
Silvia semeraro via Wikimedia Commons.
According to the Coris’ biography, Gerty’s identity as a person of Jewish heritage precluded her from receiving any university teaching positions in Europe. To better support her, and to escape the general conditions of life in Europe, the Coris decided to immigrate to the United States in 1922.
Gerty began her research career as an assistant pathologist at the New York State Institute for the Study of Malignant Diseases in Buffalo, New York, where she was eventually promoted to assistant biochemist. The Coris found increasing opposition to their collaborative relationship due to institutional nepotism policies at the time – ultimately, however, the two weathered the storm and continued their professional collaboration. During these early years, Gerty found great interest in studying the effects of X-rays on skin and body organs, which some have speculated had severe detrimental effects on her health later in life.
According to family lore, Gerty’s father was diabetic. He had said to his daughter, “Find me a cure!” – sparking Gerty’s lifelong commitment to sugar metabolism. At Buffalo, the Coris researched what exactly in the body regulated blood glucose. By studying rats, they determined that the hormone insulin increased the conversion of glucose to muscle glycogen but decreased conversion to liver glycogen. On the contrary, the hormone epinephrine decreased muscle glycogen and increased liver glycogen. Based on the known assumption that muscle glycogen did not add glucose to the blood, the Coris postulated that another intermediate would be necessary to complete the cyclic conversion of muscle to liver glycogen. They discovered that lactate was the missing puzzle piece in the “cycle of carbohydrates” – later to be known as the Cori Cycle.
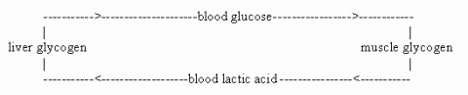
The famous Cori Cycle describing how glycogen is processed in the body
Cori and Cori, 1928
In 1931, the Coris moved to the Washington University School of Medicine in St. Louis, where they continued their work on carbohydrate metabolism. Within five years, the Coris had identified glucose-1-phosphate (later deemed the Cori ester) – the intermediate that represented the product of the first step from the conversion of glycogen to glucose.
This compound served as a jumping off point for Gerty’s burgeoning interest in how enzymes work, or enzymology. Step by step, the chemical process by which glycogen was synthesized, appeared. First came the enzyme phosphoglucomutase, which converted glucose-1-phosphate to glucose-6-phosphate (another intermediate). Then came the seminal discovery of phosphorylase, crystallized by Gerty and her graduate student Arda Green, which catalyzed glucose-1-phosphate formation from glycogen. Green would go on to co-discover the neurotransmitter serotonin, and how fireflies glow.
Aiming to synthesize glycogen, the Coris reversed the phosphorylase reaction, adding a little glycogen to push the equilibrium in the right direction. Excitingly, a starch-like macromolecule appeared – debunking the belief that cells were required to synthesize these very large molecules. It was the first time that any macromolecule had been synthesized in a test tube. This discovery, coupled with the others, launched the Coris to Sweden for the Nobel Prize. More importantly, the newfound capability to study macromolecules in a test tube formed the foundation for Gerty’s work with glycogen structure and glycogen storage diseases.
Immediately after returning from Sweden, Gerty began to turn her attention to glycogen structure and function. Glycogen appears as a branching macromolecule with various combinations of carbon linkages – like a starchy tree. Certain linkages connected the linear portions of the molecule, while another linkage type connected the branching portions. How, then, could glycogen be broken apart?
Along with her graduate student, Joseph Larner, Gerty hypothesized that an enzyme, later called debranching enzyme, could break the branch point linkages in glycogen. If the hypothesis was correct, the only product from the breakdown of glycogen by this enzyme should be glucose.
Lo and behold, when the experiment was conducted, free glucose appeared as the only product. As Joseph remembered, Gerty ran up the hallway in excitement, screaming, “It’s free glucose, it’s free glucose!”
The discovery of the debranching enzyme combined with Gerty’s increased knowledge of glycogen’s structure formed the basis for her studies on glycogen storage diseases, a group of conditions with no known cure even today. Then known as a singular disease, these conditions affected around 1 in 100,000 births. They also manifested first as a pediatric disease – marking Gerty’s return to the beginnings of her medical career, all those years ago.
_and_Carl_Ferdinand_Cori.jpg)
Gerty Cori and her husband Carl Cori were jointly awarded the Nobel Prize in medicine in 1947 for their work on how the human body metabolizes sugar
Smithsonian Institution via Wikimedia Commons
Gerty had a hunch that the disease was somehow due to a defect in glycogen-related enzymes. Her guess was that the missing enzyme was glucose-6-phosphatase – the enzyme that broke down glucose-6-phosphate. Joseph, her graduate student, on the other hand, believed that the missing enzyme was the debranching enzyme.
Gerty had saved a cabinet of patient glycogen samples, sent by physicians interested in learning more about glycogen storage diseases. It was decided that they would stain the samples using iodine to determine the structure of glycogen in this disease. Iodine stained normal glycogen to a brownish color, while staining starches to a blueish-purple color.
The stain showed a blueish-purple color – indicating that the glycogen demonstrated greater morphological similarity to starch. This represented a watershed moment in biochemistry; the stain indicated that glycogen storage disease contained glycogen with structural differences, which showed that this was a molecular disease. At the time, this marked glycogen storage disease as only the second known molecular disease.
As Gerty continued to study the disease in greater detail, she began to identify different forms of the disease. Her findings were summarized in a 1952 Harvey lecture, one of the most prestigious lecture series for biomedical science. Rather than the one-size-fits-all definition that it had originally been given, Gerty identified four main disease forms: the first, due to a lack of glucose-6-phosphatase in the liver (consistent with her original hypothesis), the second, due to a lack of de-branching enzyme (consistent with Joseph’s hypothesis), the third, due to a lack of another branching enzyme, and the fourth, with unknown enzymology that led to more generalized organ disease.
From her enzyme kinetic studies on glycogen storage diseases, Gerty unknowingly established the first set of experiments that directly linked enzyme dysfunction with disease. Before, it had just been assumed that enzymes could cause disease, but never proven. Her final paper, published the same year of her death in 1957, was a review on glycogen storage disease.

National Library of Medicine via Wikimedia Commons
During the final years of her life, Gerty refused to give up laboratory work. Instead, she requested a small cot be added to the laboratory, upon which she would rest when tired. She also held a party, according to graduate student Mildred Cohn, “to squelch the rumor that I [Gerty] was dead.” Finally, Carl was put in charge of monitoring her blood hemoglobins, eventually carrying her around when she could not stand – the final act in a marriage partnership that changed the face of biochemistry.
From Joseph’s recollections, Gerty “needed only one exciting experimental finding to jump into a problem with unbounded energy.” Her spontaneity, particularly in thinking of new ideas, manifested in the breadth of research discoveries she accomplished. Gerty was undeniably brilliant, with a legacy lasting far beyond her scientific accomplishments. Her graduate students, many of them women, went on to pursue their own careers in enzymology and metabolism.
