
Photo by Thao Le Hoang on Unsplash
Fights are good for science. Just ask Elizabeth Nolan or Louis Pasteur
Disagreements over Nolan's findings on slowing down bacteria are just part of a healthy scientific process
Harsh criticism has propelled scientific discoveries for centuries. Just look at Louis Pasteur and Robert Koch in the late 1800s — their intense rivalry as they each investigated anthrax provided the scientific evidence for germ theory. While some could argue these two scientists would have made their discoveries without their bitter rivalry, well-documented fiery clashes at international conferences drove both men back to their labs to disprove the other. In fact, the fundamental Koch’s postulates that describe the relationship between microbes and disease are now commonly taught in microbiology classes.
Lactobacillus (small rods) near a skin cell
CDC/Dr. Mike Miller
Now, another grand debate is taking place — this time, over a protein’s role in the human immune system. In 2015, Dr. Elizabeth Nolan, a chemistry professor at MIT, published a landmark paper in Nature on calprotectin. Calprotectin, a protein released by white blood cells, prevents bacteria from spreading by starving them of essential nutrients. When bacteria invade our bodies, they steal essential nutrients our bodies need, including metals such as iron. These metals make bacteria better able to infect and harm us. But our bodies have evolved ways to protect the metals we consume — for example, by releasing calprotectin. This process is sometimes called nutritional immunity.
Calprotectin was previously known to capture manganese and zinc in order to starve bacterial invaders. Nolan found that, in response to the bacterium Pseudomonas aeruginosa, the protein also captures iron. In fact, Nolan’s team monitored the way that many common metals, including manganese, zinc, copper, iron, calcium, cobalt, and nickel interacted with calprotectin. Knowing exactly how our bodies respond to different bacterial pathogens can help pharmaceutical companies design drugs to support those functions and even artificially replicate them.
That landmark 2015 paper was based on findings from one of Nolan's former graduate students, Toshiki Nakashige, who tested calprotectin against several species of bacteria in the lab. Nakashige found that the bacteria that required iron for growth grew more slowly when calprotectin was added to the growth mixture. The presence of calprotectin inhibited iron uptake in two particular kinds of bacteria, E. coli and P. aeruginosa, by a factor of three.
Nolan and her collaborators also took a close look at the unusual metal-binding site in calprotectin itself. They have observed that calprotectin chelates, or binds to, iron in a very unusual way. But not everyone agrees with Dr. Nolan's findings. Until 2015, previous studies focusing on calprotectin’s metal interactions had found that the protein does not interact with iron at all. These studies, in high-impact journals like PNAS and Science, were published by Dr. Eric Skaar and Dr. Walter Chazin of Vanderbilt University, respectively.
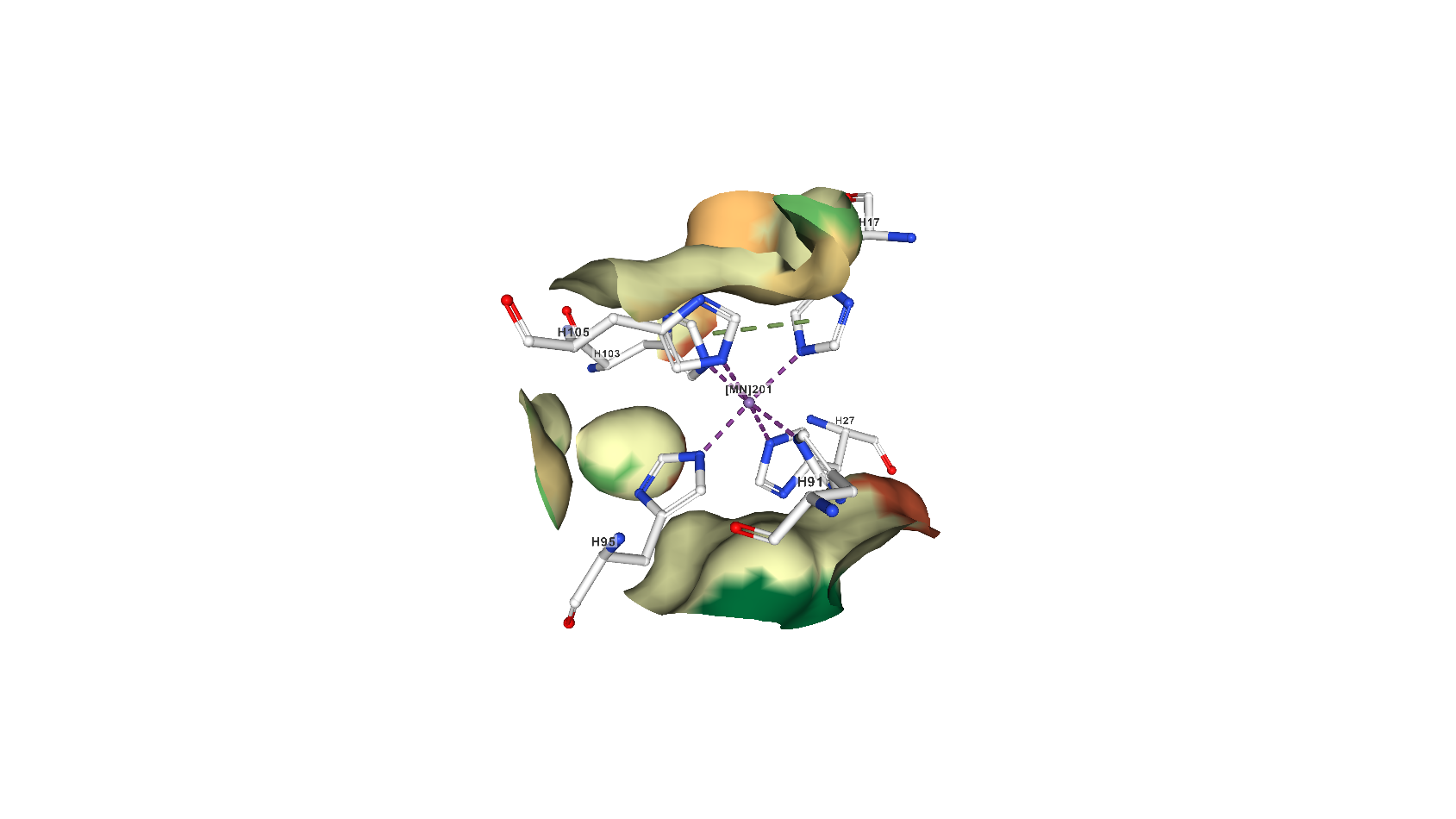
The pocket where calprotectin holds on to metals, in this case a single manganese atom, seen as a purple sphere in the center.
PDB 4GGF, Damo et al 2013 Proceedings of the National Academy of Sciences
Each of the research labs used a variety of methods to arrive at their differing conclusions. Nolan's team used ICP-optical emission spectra. To confirm their findings, they used Mossbauer spectroscopy. This incredibly sensitive technique uses gamma rays to show how an atom's energy level shifts, which occurs during binding, such as when iron atoms is chelated by a protein.
In contrast, Shaar and Chazin used either equivalent or more sensitive techniques in their studies. Furthermore, Shaar and Chazin's studies in mice to date have not demonstrated any iron chelation activity or iron-chelation antimicrobial activity for calprotectin.
Nolan acknowledged in her 2019 follow-up paper that her contradictory conclusions “sparked some debate in the literature.” And much like Koch and Pasteur, this debate is shaping up to be among big names in big camps, pitting MIT’s prestigious chemical biology lab against the foremost microbiology program in the country at the University of Illinois at Urbana-Champaign and a long-established medical research program at Vanderbilt.
But unlike Koch or Pasteur, who in response to criticism wrote scathing replies attacking (but not empirically disproving) the others’ theories, Nolan's work shows that she has carefully evaluated the two strongest critiques against her novel discovery: the relationship to iron, and the role of calprotectin in nutritional immunity. When Dr. Thomas Kehl-Fie's research group published experimental results in opposition to Nolan's findings, clearly skeptical of her claims on both calprotectin's ability to bind iron and the purpose of that function, Nolan determined that their conclusions differed because Kehl-Fie's group used different growth environments for the same bacteria.
Nolan has thoroughly evaluated the counter-claims to her novel discovery and even offered alternative explanations for discrepancies. In contrast, at the moment it appears her critics dismiss the “unusual” conclusions of her studies due to their incredible nature, rather than faults in her experimental design.
Scientists have long lived and died by the (empirical) strength of their work. But when a scientist is told they are wrong, they choose their reaction. Some, like Isaac Newton, have tried to get their critic removed from notable professional societies; others have organized entire conferences and encourage presenters of opposing opinions to present (for example, Alfred Wegener and the American Association of Petroleum Geologists). In response to her critics, Nolan has demonstrated how to professionally respond to naysayers, furthering our knowledge of calprotectin’s unusual iron-capture strategy and likely role in nutritional immunity.




This article is a very good example of “fights” in science. The scientific process relies on healthy arguments between researchers. I especially love this quote: “In response to her critics, Nolan has demonstrated how to professionally respond to naysayers…” Learning how to respond to our critics make us all better scientists! It is in this criticism that can help us strengthen our research and discover amazing things. Really great read Lauren!