
Illustration by Allan Lasser
Scientists are gluing teeth to power saws to learn how sharks eat
The cutting edge of science isn't always high-tech
Katherine Corn spent the summer of 2014 using epoxy to glue shark teeth to a saw blade. Her research methods were primitive by most standards, including those of her classmates at Friday Harbor Labs (FHL), where the then-Cornell University undergrad (now a grad student at UC Davis) was taking a summer course on fish biology. Yet, with the encouragement of instructor Adam Summers and teaching assistant Stacy Farina, Corn was determined to build a simple but more realistic way to simulate the dramatic thrashing of a shark’s body as it feeds.

Corn was interested in sharks because she knew that their behavior impacted hundreds and thousands of other species. Thanks to marine laboratories like FHL, scientists have realized that the rules governing ecosystems in the ocean are the same as those governing ecosystems on land. Take the idea of keystone species.
A keystone species is one whose presence in an ecosystem shapes the entire community. On land, a famous example is the Yellowstone grey wolf, whose reintroduction to the national park in 1994-1995 has kept elk populations in check, preventing overgrazing. Less grazing has allowed new aspen forest growth, and since these forests are home to many other animals, like beavers, the populations of these animals has grown, too.
In the ocean, sharks are a keystone species, shaping communities in the same way the wolves do in Yellowstone. Sharks keep other animals in an ocean ecosystem in a healthy balance. Overfishing, coupled with their slow growth and reproduction, have left many shark species endangered, threatening the role they play in balancing ecosystems. That’s why scientists like Corn, Farina, and Summers are interested in what exactly sharks are doing in the open water.
The problem is, observing sharks in the wild is difficult. It can take days at sea just to reach them. Once there, scientists must rely on expensive camera-equipped underwater vehicles to be their eyes, ears and hands underwater.
Instead, the researchers wanted to use something more available, the sharks’ teeth, to investigate a fundamental question about their feeding behavior: how does a predatory shark eat?
.gif)
The shark-attack videos you find in TV documentaries suggest a simple answer: a fast, surprising strike at a prey item, where the shark hits the food hard and makes a straight puncture with its teeth. Thrashing ensues as the shark shakes its head, slicing out a mouthful of food, before the shark disappears again. But, like many things, shark feeding turns out not to be so simple.
Walk into any natural history museum, or spend enough time looking at pictures of jaws and skulls of animals on the internet, and you will find that you can make some solid guesses about what a mammal eats based on its teeth. Long, pointy teeth? Carnivore. Flatter, broader teeth with parallel grooves? Herbivore. In between? Omnivore. In all three cases, however, the teeth can be categorized as incisors, canines, and molars, each with different functions—slicing, shredding, and crushing, to name a few.
What documentaries broadcast during shark week fail to portray is that the tooth diversity in sharks rivals that in mammals. In fact, the details of tooth shape can be so specific that they're often used as a tool to identify shark species. Shark teeth can be spear-like, simply triangular, notched, or multi-cusped. Unlike in mammals, though, it's unclear why the diversity in shark teeth exists. What do each of these tooth types actually do?
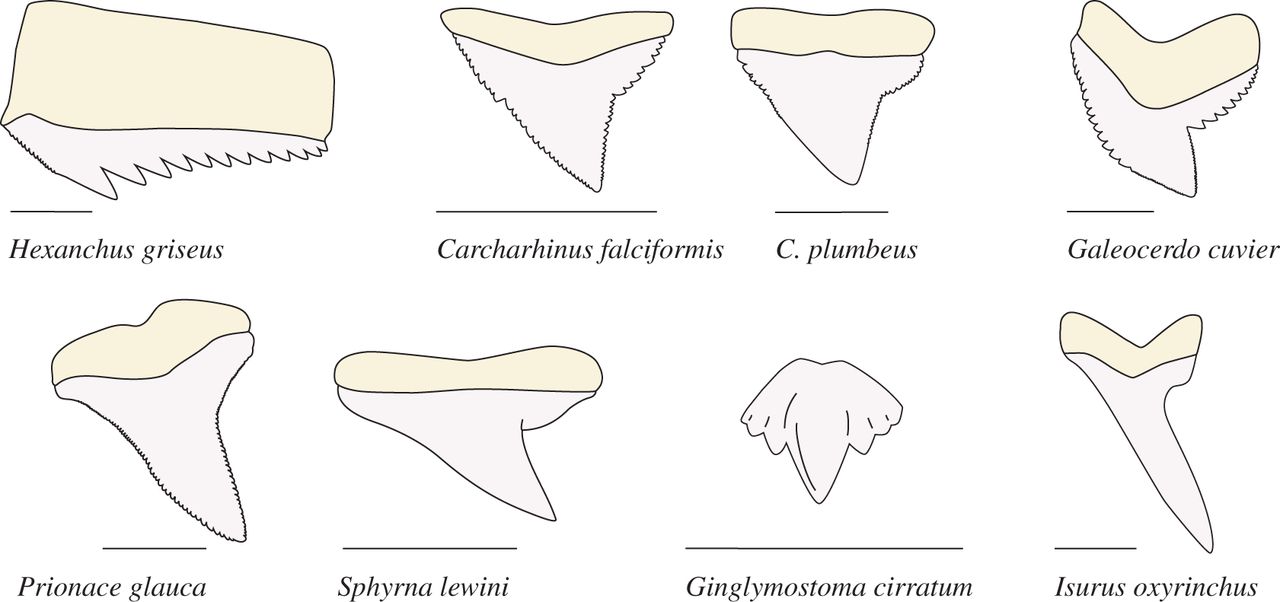
The answers to this question are incredibly important. Being able to predict what an animal eats based on its teeth helps us understand how the animal fits into the food web. Understanding food webs helps us to understand the inner workings of an ecosystem, and how it may be affected by the loss or gain of a particular animal. This information can provide us with a view of contemporary ecosystem health, but the abundance of tooth types in the fossil record also can provide insight into the food webs of the far past and how they have evolved into the ecosystems we see today.
Identifying shape-function relationships in shark teeth is not a new idea. In a 2010 study, Lisa Whitenack and Philip Motta, then at the University of South Florida, placed shark teeth in a materials testing system, a standard device used by engineers to study material properties like tear, stretch, and fracture resistance. With this machine, they could try to cut a handful of materials using each tooth, and compare how well each tooth punctured or sliced each material. The trouble was, none of these studies found any difference between the cutting abilities of the different types of shark teeth. Was the theory flawed? Maybe no clear-cut relationship like we see in mammals existed in shark teeth after all.
Undeterred, Corn decided to make one more attempt to find out at FHL. Together with Summers and Farina, she had a revelation: the fancy engineering equipment used in prior studies, while great for understanding the properties of tooth material, wasn’t putting shark teeth in a realistic environment. The sophisticated equipment simply could not match the speed of the deadly shark head-shake we’ve all seen on TV. But, she realized, there was something that could mimic that motion in a precise, repeatable way: power tools.
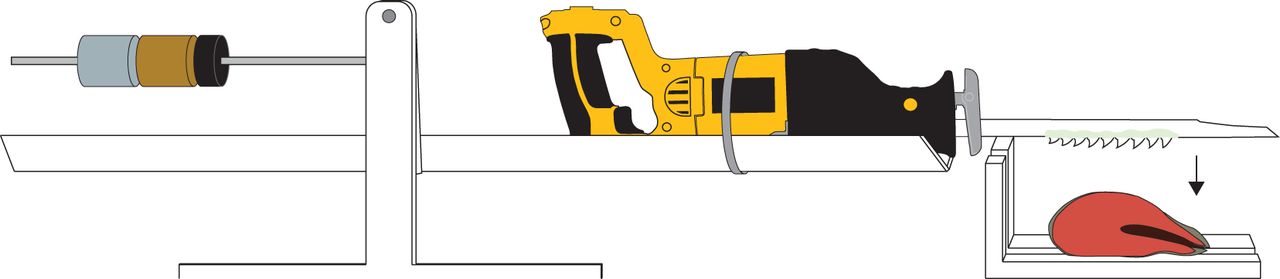
Hacking Together a New Shark Tooth Simulation
With the help of Jeffrey Brash of Valley Steel and Stone, a construction company located on the island, Corn built a new testing system to mimic shark head-shake motions more accurately. To build this system, Corn and Brash first glued shark teeth onto smooth metal blades. Then, they mounted the metal blades onto a reciprocating saw—a power tool that is normally used to move a tooth saw blade back-and-forth to cut through wood. These fast, dynamic motions allowed Corn to mimic a shark’s head-shake better than ever before.
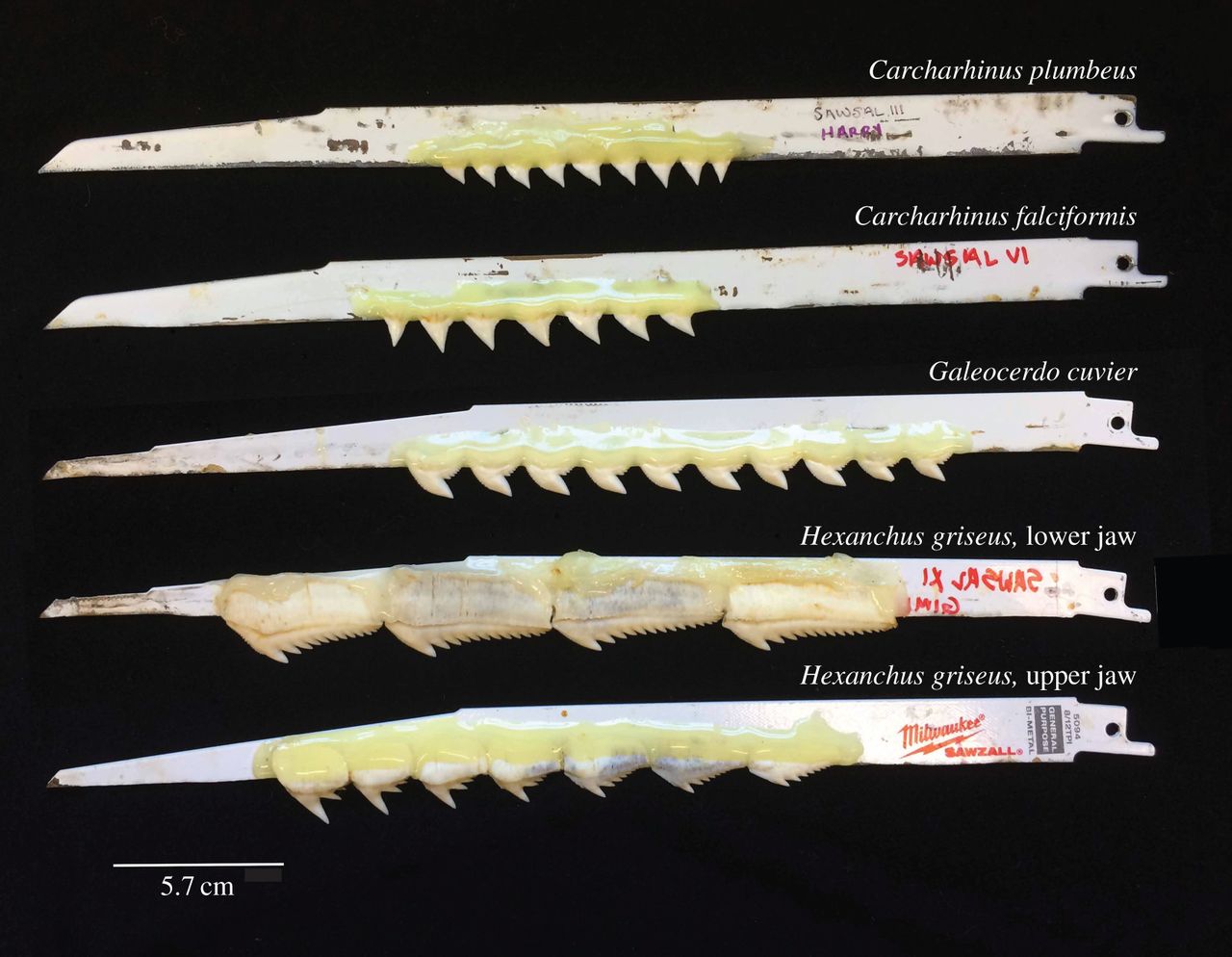
The scientists tested teeth from three main shark species representing several general shark teeth types: silky, tiger, and sixgill. The silky shark teeth are nearly triangular, the tiger shark teeth are notched and look like triangles with a bent tip, and sixgill sharks are multi-cusped, featuring multiple points.
Slow-motion video of a blade with teeth from Carcharhinus falciformis on its second use.
Full-speed video of a blade with teeth from Hexanchus griseus on its fifteenth use.
To control the “bite force” of the saw, the researchers developed a seesaw-like holder. The shark saw was placed on one end, and counterweights on the other, so that the saw blade could cut a piece of grocery-store salmon using a small, controlled amount of force that was consistent between trials. While the saw did its work, the researchers filmed it using a high speed camera, and used still images from the footage to measure cut depth and length. The measurements were then combined into a cutting ability metric: the “area” (depth times length) of a cut made in each oscillation per tooth width.
Corn and her team conducted two types of tests. First, they tested how each tooth type cuts initially by running multiple shark tooth blades from each species through raw salmon. Second, they ran a single shark tooth blade from each species for about twenty tests in a row to see how quickly repeated use made a tooth dull.
Life-Like Models Yield Promising Results
Unlike earlier, simplified tests of shark tooth function, the new dynamic tests using the shark saw found statistically significant differences between some of the shark teeth. Silky and tiger shark teeth had three to six times more cutting ability than the sixgill shark teeth. However, the silky and tiger shark teeth both dulled quickly, while the sixgill teeth did not dull much at all.
Corn and her colleagues didn’t run further experiments to explain these differences. But they hypothesize that the ability of each of these sharks to replace teeth may be behind the observations. The sixgill shark, which lives in cold water and has a lower metabolic rate, cannot replace its teeth as fast as tiger or silky sharks, who live in warmer water. As a result, sixgill sharks may eat softer prey with more blunt teeth that dull slower. Because they can replace them quickly, tiger and silky sharks may not have to worry about dulling their sharp teeth with harder prey that must be cut into pieces, like the sea turtles tiger sharks are known to eat.
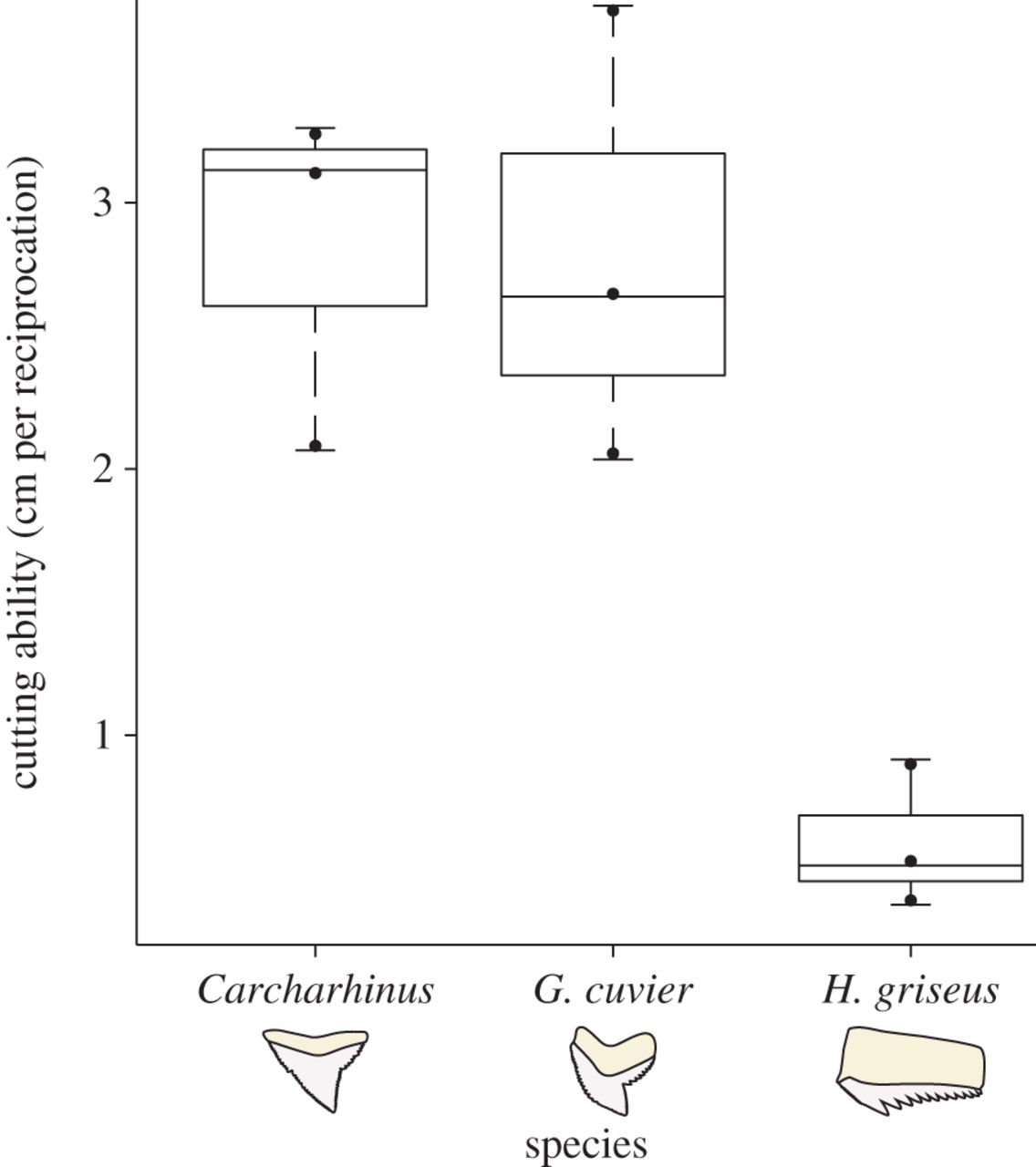
Corn’s study was quite small—investigating three of around 400 known species—and the results shouldn’t be applied too widely. Nonetheless, her team’s findings represent some of the first experimental data demonstrating that shark tooth types may have different functions. They also show how little we understand about shark feeding and tooth function, despite our obsession with shark documentaries.
Corn also proved that sometimes, simpler can be better. Despite their ability to precisely replicate experiments, the specialized material testing equipment used in previous studies may not have realistically modeled shark feeding behavior. This shortcoming could have masked important differences in shark tooth function, making it harder to understanding of the key roles that sharks play in the ocean environment.
Produced with the Derek Bok Center for Teaching and Learning at Harvard University. Learn more about how we work with partners.
Featured Article
Corn KA, Farina SC, Brash J, Summers AP. 2016 Modelling tooth–prey interactions in sharks: the importance of dynamic testing. R. Soc. Open Sci. 3: 160141. http://dx.doi.org/10.1098/rsos.160141
Other References
- Cabrera-Chávez-Costa AA, Galván-Magaña F, Escobar-Sánchez O. 2010 Food habits of the silky shark Carcharhinus falciformis (Müller & Henle, 1839) off the western coast of Baja California Sur, Mexico. J. Appl. Ichthyol. 26: 499–503. http://dx.doi.org/10.1111/j.1439-0426.2010.01482.
- Ebert DA. 1994. Diet of the sixgill shark Hexanchus griseus off southern Africa. S. Afr. J. Mar. Sci. 14: 213–218. http://dx.doi.org/10.2989/025776194784287030
- Farina, Stacy C. Personal interview. 3 Mar. 2017.
Heithaus MR, Frid A, Wirsing AJ, and Worm B. 2008. Predicting ecological consequences of marine top predator declines. Trends Ecol. Evolut. 23: 202-210. http://dx.doi.org/10.1016/j.tree.2008.01.003 - Lowe CG, Wetherbee BM, Crow GL, Tester AL. 1996 Ontogenetic dietary shifts and feeding behavior of the tiger shark, Galeocerdo cuvier, in Hawaiian waters. Environ. Biol. Fish 47: 203-211. http://dx.doi.org/10.1007/BF00005044
- Motta PJ, Huber DR. 2012 Prey capture behavior and feeding mechanics of elasmobranchs. In Biology of sharks and their relatives (eds. Heithaus MR, Carrier JC, Musick JA) 153–209. Boca Raton, FL: CRC Press.
- Purdy RW. 2006. A key to the common genera of Neogene shark teeth. Smithsonian National Museum of Natural History. Accessed: 28 Feb 2017. https://paleobiology.si.edu/pdfs/sharktoothKey.pdf
- Ripple WJ and Beschta RL. 2012. Trophic cascades in Yellowstone: the first 15 years after wolf reintroduction. Biol. Conserv. 145: 205-213. http://dx.doi.org/10.1016/j.biocon.2011.11.005
- Sibert E, Norris R, Cuevas J, Graves L. 2016 Eighty-five million years of Pacific Ocean gyre ecosystem structure: long-term stability marked by punctuated change. Proc. R. Soc. B 283: 20160189. http://dx.doi.org/10.1098/rspb.2016.0189
- Whitenack LB, Motta PJ 2010 Performance of shark teeth during puncture and draw: implications for the mechanics of cutting. Biol. J. Linnean Soc. 100: 271–286. http://dx.doi.org/10.1111/j.1095-8312.2010.01421.x
- World Wildlife Fund. 2017. Shark. WWF.org. Accessed: 27 Feb 2017. http://www.worldwildlife.org/species/shark
