
3D-printed organs could help us heal faster and live longer
Researchers made waves last year by successfully implanting new jaws in rats
Three-dimensional printing is a hot technology right now, but scientists have struggled to use it to solve a pressing problem: helping people heal. Healing wounds is a tall order for even the best bioprinters - creating materials that can meet the body's needs for repair and regeneration and also get printed is no small feat. But experiments keep coming closer to making it work, putting the exciting possibility of a 3D-printed transplant future squarely in scientists' sights.

The printer used to create the implants.
Wake Forest University
Cells require specialized conditions to grow and develop, but to pass through a 3D printer, these materials also need to be smooth enough to keep these specialized 'bioprinters' from jamming. The printers also need to make material that is strong enough to support complex structures like tissues. Scientists have made stronger materials that approximate bone using plastic, but scientists had trouble getting enough cells to attach to the bone-like material to grow and form tissues. To get enough cells to form a tissue meant that scientists needed to spread a thick slather of them onto bioprinted materials: a new approach was needed so they could be spread thickly enough to flourish.
To tackle these challenges, a team of biomedical engineers at Wake Forest led by Hyun-Wook Kang (now at Ulsan National Institute of Science and Technology in Korea) developed a specialized bioprinter for a study published last year that was designed to push past traditional barriers to make tissues and organs that worked and could be integrated into a living being.
The researchers faced the challenge of creating a matrix that simultaneously contained bone, blood vessel scaffolding and cells. They also needed to make sure that enough cells were evenly printed over the tissue.
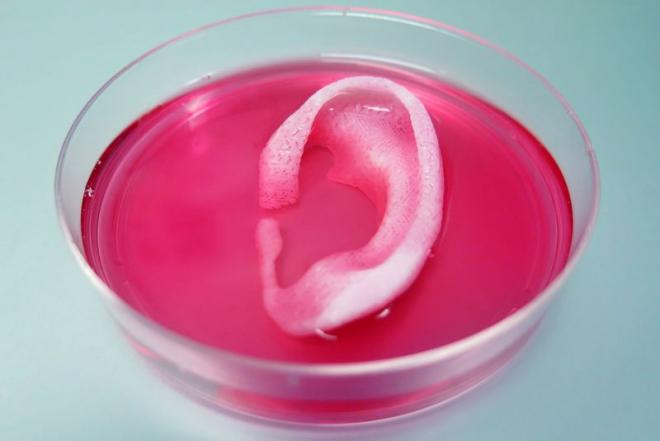
An example of a 3D-printed ear sitting in medium
Wake Forest Institute for Regenerative Medicine
Bioprinters are like souped up 3D printers. Rather than mimicking the physiological conditions of cells and tissues, they instead "print" layers of material – one on top of each other – which create shapes that form building blocks for bone and muscle growth.
The scientists created a custom cocktail of plastic and calcium as a base for the ink, and then they added a bit of soap to keep things from jamming up – an everyday remedy for a complex problem. They could simply rinse out the soap once the material had dried solid. The bioprinter used two different types of printing to make different types of material: a more dense, bone-like material, and a weaker muscle-like material to support cell growth. The weaker lattice was supported by a thick mold to strengthen the structure during printing.
To see if their new technology could potentially help regrow tissue, the team decided to create an implantable jaw and test if it could fix a genetic bone defect. Using one pattern of bioprinted material for the jawbone and another pattern for the surrounding muscle, the scientists printed human amniotic fluid-derived stem cells in a high-sugar jelly and polymer mixture into each jaw and allowed them to grow. The printed jaws were alive, but could they regrow actual bone?
The team put the printed jaws to the ultimate test: implantation into living rats suffering from natural jaw deformations. Would the rats reject the printed jaw, or would their bodies allow the implants to integrate into bone structure?
Five months later, the team inspected the bioprinted implants. Not only had they joined the jaw tissue, but they had grown bone to replace the defect, becoming strong and full of blood vessels, much like a healthy jaw. Jaws without the bioprinted implants formed scar tissue instead. Clearly, the bioprinted implants had worked.
These findings are exciting and pave the way for a future where we could conceivably print entire organs and tissues. Sometime within the next 10 years, we may rely on bioengineers to replace failing organs rather than relying on transplants, and enter a future where bionic organs can help people lead longer, healthier lives. More complex organs may need special cells and more time to grow; as usual, more experiments are needed.
